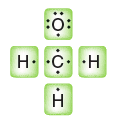
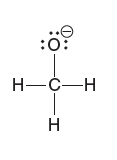
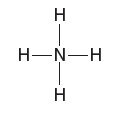
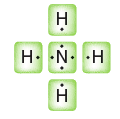
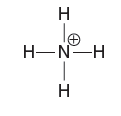
Partial charges can be visualized with three-dimensional, rainbow like images called electrostatic potential maps. As an example, consider the electrostatic potential map of chloromethane.
In the image, a color scale is used to represent areas of 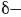 and
and 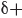 . As indicated, red represents a region that is
. As indicated, red represents a region that is 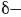 , while blue represents a region that is
, while blue represents a region that is 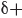 . In reality, electrostatic potential maps are rarely used by practicing organic chemists when they communicate with each other; however, these illustrations can often be helpful to students who are learning organic chemistry. Electrostatic potential maps are generated by performing a series of calculations. Specifically, an imaginary point positive charge is positioned at various locations, and for each location, we calculate the potential energy associated with the attraction between the point positive charge and the surrounding electrons. A large attraction indicates a position of
. In reality, electrostatic potential maps are rarely used by practicing organic chemists when they communicate with each other; however, these illustrations can often be helpful to students who are learning organic chemistry. Electrostatic potential maps are generated by performing a series of calculations. Specifically, an imaginary point positive charge is positioned at various locations, and for each location, we calculate the potential energy associated with the attraction between the point positive charge and the surrounding electrons. A large attraction indicates a position of 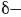 , while a small attraction indicates a position of
, while a small attraction indicates a position of 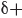 . The results are then illustrated using colors, as shown.
. The results are then illustrated using colors, as shown.
A comparison of any two electrostatic potential maps is only valid if both maps were prepared using the same color scale. Throughout this book, care has been taken to use the same color scale whenever two maps are directly compared to each other. However, it will not be useful to compare two maps from different pages of this book (or any other book), as the exact color scales are likely to be different.